What is Terrien’s Marginal Degeneration?
Surprisingly little is known about what causes Terrien’s marginal degeneration (TMD). We do know that: it’s rare, three in every four people affected are men; its onset is usually in people’s third-to-fifth decade of life, and both eyes are affected most of the time.
We know more about the pathology: the disease is characterized by thinning of the margin (edges) of the cornea where it meets the white part of the eye (the sclera). The thinning usually starts in the top part of the margin, it usually progresses and involves the whole circumference of the corneal periphery. The disease typically results in the growth of new blood vessels – neovascularization – plus scarring and lipid deposits (usually seen as fine, yellowish punctate stromal opacities) at the margin. As the degeneration progresses, it usually leads to increasing against-the-rule or oblique astigmatism. The cornea can become severely weakened, spontaneous ruptures of Descemet’s Membrane can occur and result in interlamellar fluid or corneal cysts. Although spontaneous perforation is rare, even minor trauma can perforate the cornea. The question is: how can TMD be treated?
Until recently, there wasn’t much that could be done, other than trying to manage the patients’ astigmatism with spectacles or rigid gas-permeable lenses and careful monitoring of corneal thinning. But if the cornea becomes so thin that perforation is imminent, then corneal transplantation is then the logical next step – although given the fact that the disease is peripheral in nature, and the corneal transplants have to extend to the periphery of the cornea… successful treatment can be extremely hard to achieve.
However, there is another approach that might help.
Can Terrien’s Marginal Degeneration be cross-linked?
Corneal cross-linking (CXL) can successfully treat corneal ectasias (thinning diseases) like keratoconus, post-LASIK ectasia and corneal melting. Cross-linking the proteins (mostly collagen) in the corneal stroma is thought to not only strengthen the cornea, but also render it more resistant to enzymatic digestion. If CXL can stop corneal melting (a hallmark of TMD), could it also stop TMD from progressing?
Yes. We have successfully treated several patients with TMD using CXL, applied to the area of maximal corneal thinning, and by using a sponge to cover and protect the corneal limbus from UV-illumination. We have found that CXL using the gold-standard Dresden Protocol not only improves these patients’ corneal shape, visual acuity and quality, but also reversing the corneal thinning that occurred in the melted, peripheral ectatic areas of these patients’ corneas.
The best explanation for these results is that CXL increases the resistance of the corneal stroma to enzymatic digestion. Under normal conditions, there is homeostasis – a fine balance – between corneal collagen synthesis and breakdown. It appears that CXL, by making collagen breakdown harder to occur, shifts the balance towards synthesis, which would explain the improvements in these patients’ corneal thickness seen to date. Successful treatment with CXL has the additional benefit of avoiding the need for keratoplasty – helping preserve scarce corneal tissue, and sparing the patient from the risks and demanding post-procedural eyedrop regimen that’s an integral part of corneal transplantation procedures.
These results also imply another use of CXL in TMD: prophylaxis. Unlike keratoconus in adults, which starts and stops progressing, and where CXL is only indicated for use once progression is observed, TMD tends to continue to progress inexorably. CXL performed early in the disease course of TMD could help arrest the progression of the disease early, preserving vision and perhaps sparing people from needing to use rigid gas permeable lenses or some of the later-stage manifestations of the disease.

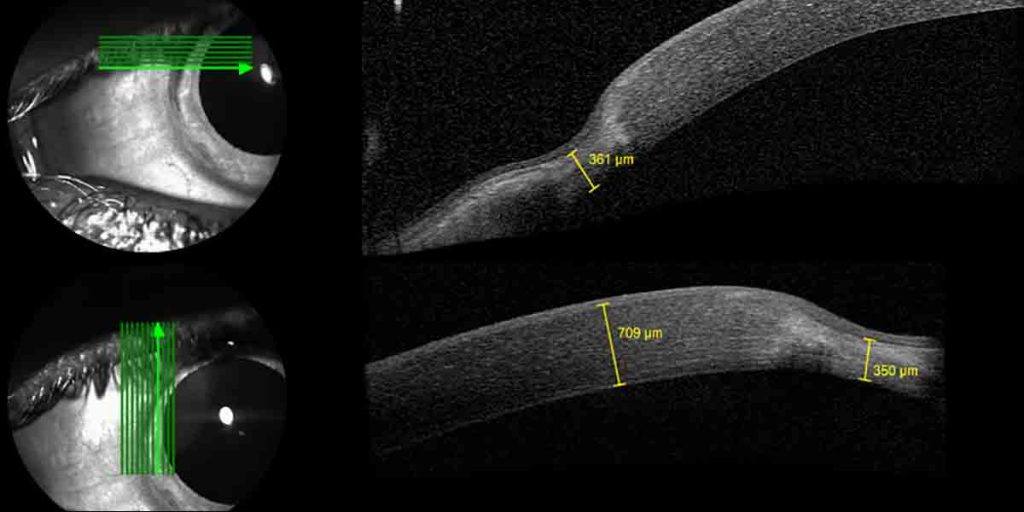
This OCT image shows a thinning of the cornea at the periphery (the edges) of the cornea)

This corneal topography map shows that the patient’s TMD has resulted in significant corneal asymmetry and therefore astigmatism.